Regulating autophagy pathway to promote clearance of protein aggregates
Project leader: Rubén Gómez Sánchez
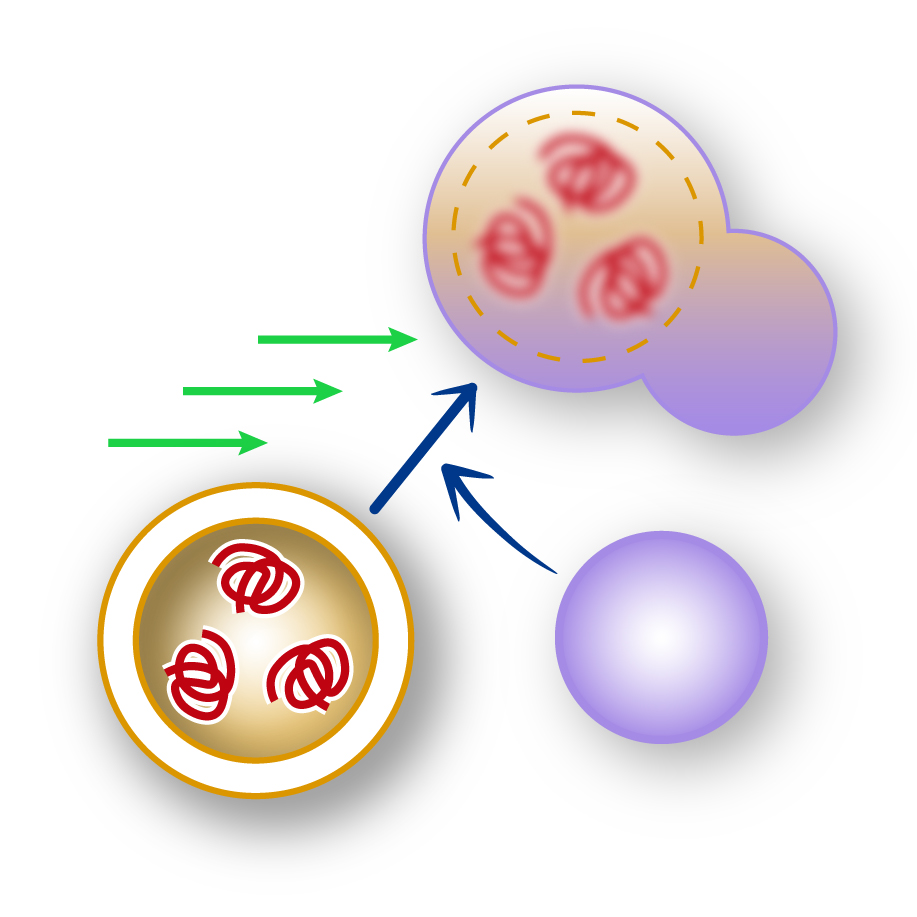
Autophagy is a highly conserved process for degrading and recycling unwanted and potentially toxic cellular components, including protein aggregates that accumulate in various disease states. The relevance of autophagy in human pathologies has emerged with the discovery that an impairment or defect in autophagy can cause diseases such as cancer or neurodegenerative disorders, where misfolded proteins form toxic aggregates that disrupt cellular function. Crucially, modulation of autophagy has been shown to be used as a therapy to delay the onset of specific diseases, including muscular dystrophies and neurodegeneration, by enhancing the clearance of these pathological protein deposits.
Autophagy is regulated by specific players, the so-called Atg proteins, but despite their relevance, their precise molecular role in protein aggregate clearance is still largely unknown. The goal of this research is to study how these players control the selective recognition and degradation of protein aggregates through the autophagy pathway. For this purpose, we take advantage of the budding yeast as a model system because of its amenability to genetic and biochemical approaches, while also validating our findings in human cell lines to ensure translational relevance. Importantly, autophagy operates in yeast almost identically as in human cells, making it an ideal system for mechanistic studies.
Our research combines yeast and human cell models to investigate how autophagy can be regulated to promote the efficient clearance of disease-associated protein aggregates. The ultimate aim of these investigations is to provide the knowledge that will help manipulate autophagy pathways to enhance protein aggregate clearance for the benefit of human health, potentially offering new therapeutic strategies for protein misfolding diseases.